Inventions Printable Worksheets - are a functional source for both understanding and company. They deal with different demands, from educational activities for kids to planners and trackers for adults. Whether you're teaching math, language, or science, printable worksheets give organized advice to boost understanding. Their customizable style enables you to tailor material to individual goals, making them ideal for educators, pupils, and specialists alike.
These templates are additionally excellent for developing appealing tasks in the house or in the classroom. Easily obtainable and printable, they conserve time while advertising imagination and efficiency. Explore a wide variety of designs to fulfill your unique requirements today!
Inventions Printable Worksheets
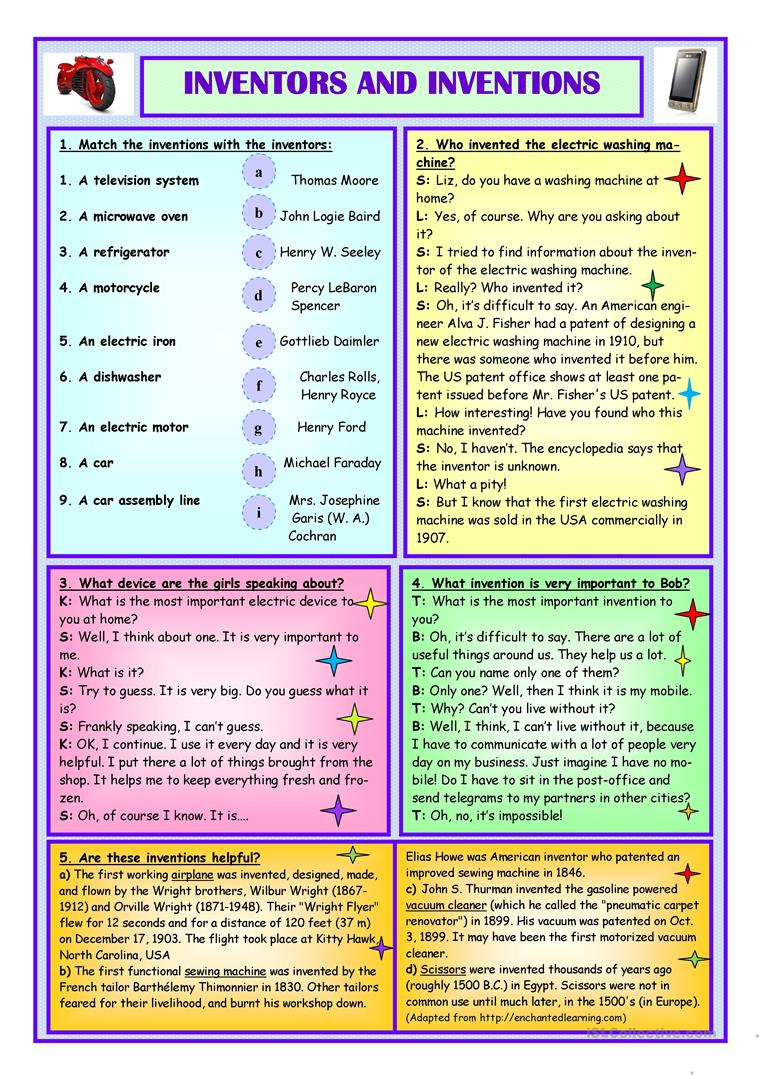
Inventions Printable Worksheets
Math teachers teach students geometry measurements money and algebra for the fifth grade curriculum Students have instant help to learn the math skills needed for their assignments Teachers show students how to use the scratchpad to analyze and breakdown math problems With Math Games, students can work at a suitable level for their individual abilities, in a format that’s simple to use and lots of fun to engage with! They can also use our digital textbook, game applications or PDF worksheets. Choose a skill to start playing.
Grade 3 Worksheets Practice With Math Games

Thomas Edison Printables For Kids
Inventions Printable Worksheets3rd Grade Math - Develop multiplication and division strategies within 100. Understand unit fractions with a numerator of 1, rectangular area and analyzing shapes. Math Games offers online games and printable worksheets to make learning math fun Kids from pre K to 8th grade can practice math skills recommended by the Common Core State Standards in exciting game formats
Math Games are free online games that help you practice math and learn new skills at the same time. Dive into an engaging game experience tailored to your individual skill level. Inventions Printable Worksheets Peggy Worksheets Grade 3 Ancient Inventions Writing Worksheet
Algebra Practice With Math Games

Inventions Worksheet For Fifth Grade
Math Games Practice Skills Skills by Standard Skills by Grade Skills by Category Math Tips Teachers Skills by Standard Kindergarten Worksheets Counting K 1 Count Objects Up to 100 K 2 Count Using Grouped Objects Up to 100 K 3 Count Using Multiple Choice Objects Up Black Inventors Worksheets
Complete our free worksheets to refresh and review their knowledge of a skill before learning more about it at school Love learning new math skills as they improve them by playing fun games Choose one of the skills above to get started Inventions Printable Worksheets 159 Lyana Worksheets Inventions Printable Worksheets Lexia s Blog

Passive Inventors And Inventions Worksheet Free Esl Printable

Inventions Printable Worksheets 159 Lyana Worksheets

Inventors And Inventions ESL Worksheet By Turac
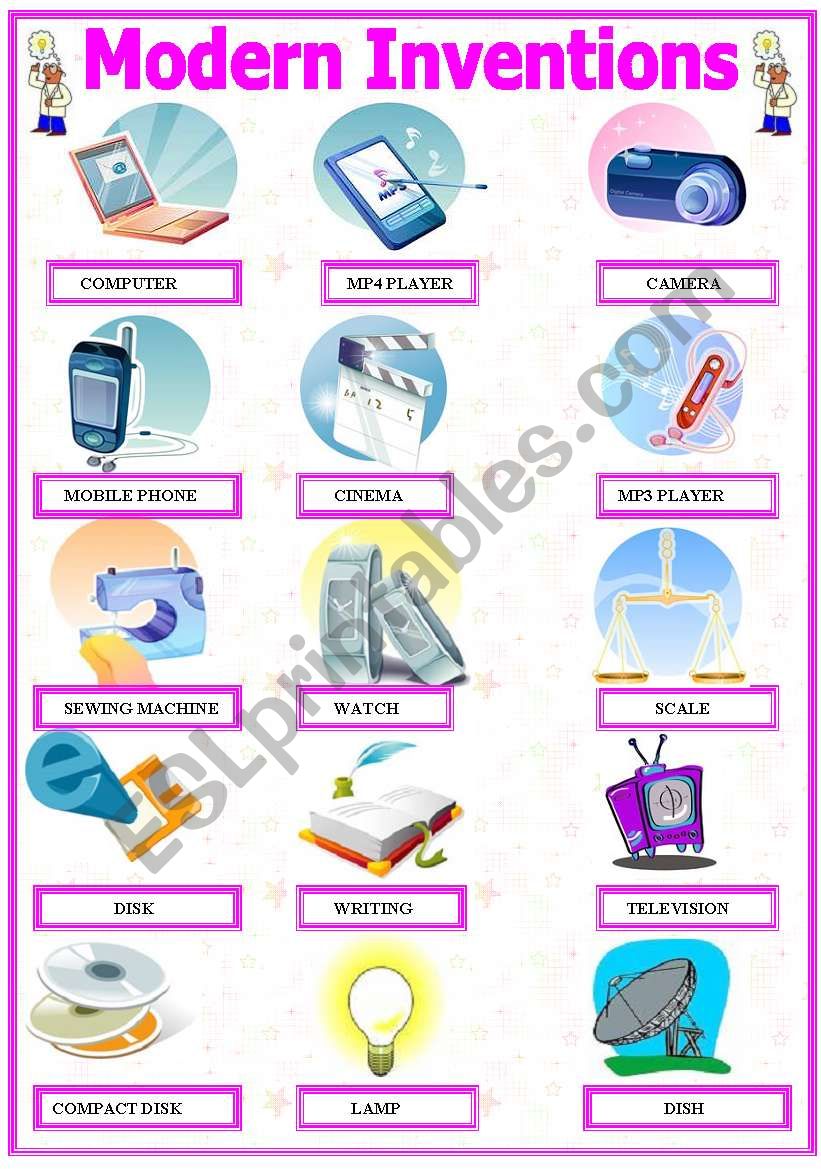
Invention Worksheets For Kids

Inventions Printable Worksheets Peggy Worksheets

What An Invention Worksheet Free Esl Printable Worksheets Made

Inventions Printable Worksheets Lexia s Blog

Black Inventors Worksheets

Inventions Worksheet For Fifth Grade

Free Printable African American Inventors Worksheets