Free Printable Us State Worksheets - are a versatile resource for both knowing and company. They accommodate various needs, from educational activities for kids to planners and trackers for grownups. Whether you're educating mathematics, language, or science, printable worksheets offer structured guidance to improve understanding. Their personalized layout enables you to tailor material to private goals, making them optimal for instructors, trainees, and experts alike.
These templates are also excellent for creating appealing tasks in the house or in the class. Quickly obtainable and printable, they conserve time while advertising imagination and efficiency. Explore a wide range of layouts to meet your unique needs today!
Free Printable Us State Worksheets

Free Printable Us State Worksheets
Once you determine a separate required minimum distribution from each of your traditional IRAs you can total these minimum amounts and take them from any one Calculate your distribution amount: Divide Line A by Line B3. This is the required minimum distribution amount for this account that must be withdrawn by ...
Ret required minimum distribution worksheet pdf BNY

Free US States Worksheets PDF Printable Coloring Pages
Free Printable Us State WorksheetsUse our required minimum distribution (RMD) calculator to determine how much money you need to take out of your traditional IRA or 401(k) account this year. Your required minimum distribution RMD worksheet Calculate and track RMDs for all your tax deferred retirement accounts Learn the basics below and then
Depending on your client's circumstances, the Worksheet for Owners or the Worksheet for Beneficiaries will calculate the RMD based on the data you enter. note. US State And Regional Worksheets ESL ELL Newcomer Made By Teachers Free Printable 50 Us States Worksheets For Kids Artofit
IRA DISTRIBUTION CALCULATION WORKSHEET Northern Trust

US Map Coloring Pages Best Coloring Pages For Kids United States
1 Enter the year end balance of your Traditional IRA in the RMD worksheet below This balance must be as of December 31st of the prior year States And Capitals Worksheets State Capitals Worksheet States And
Use this worksheet to figure this year s required withdraw for your traditional IRA UNLESS your spouse1 is the sole beneficiary of your IRA and he or she is Free United States Coloring Maps Map United States Coloring Pages Whether You Are Learning About US States In Class Or Taking A Family

Map Of The Us States With The Capitals Printable Printable Map Of The US
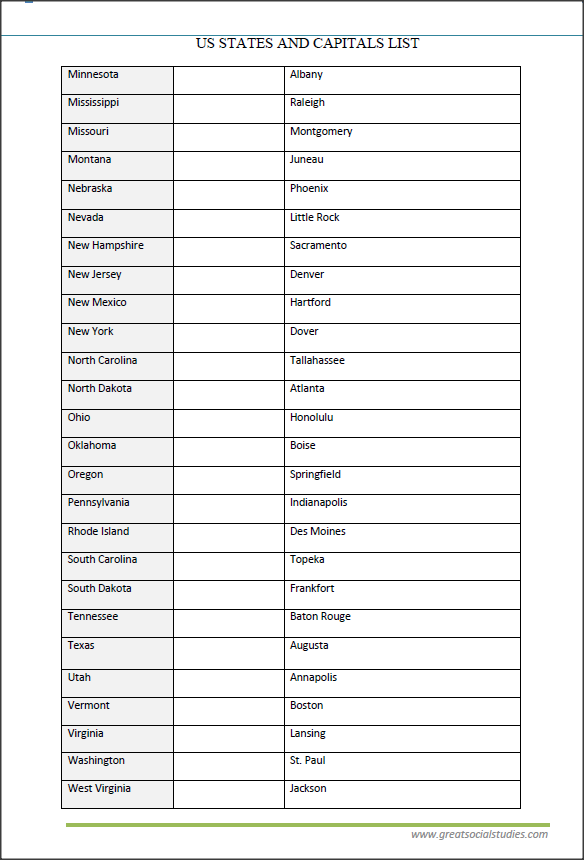
Free US State Printable Worksheets Worksheets Library

Perfect State Abbreviations Printable And Worksheets Easy To Print
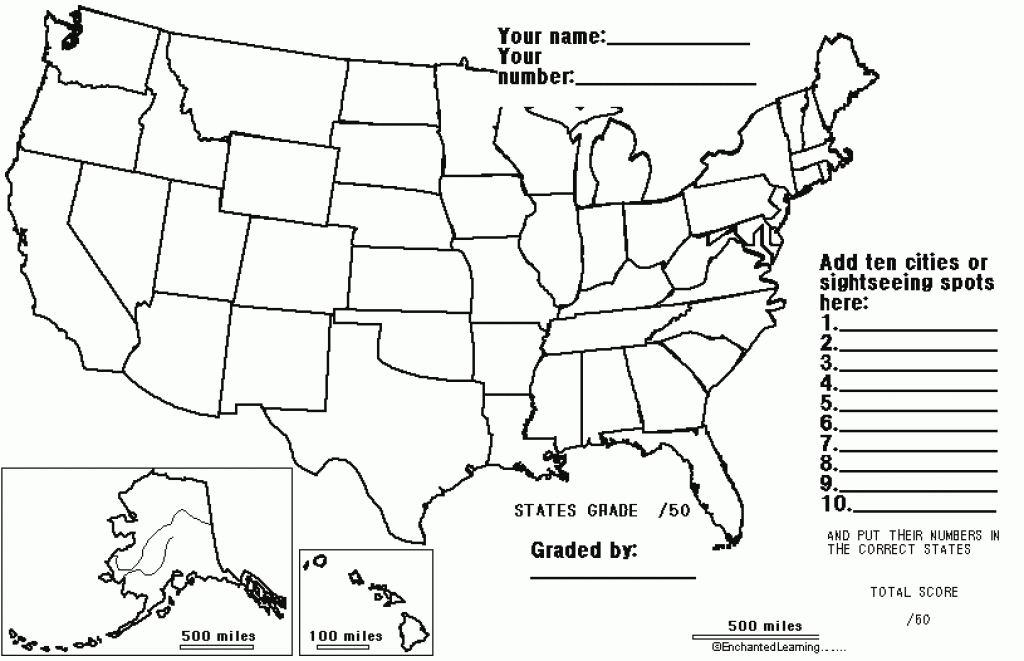
Blank State Map Quiz Printable Map

26 Best Ideas For Coloring Us Map With Capitals Printable Map Of The US
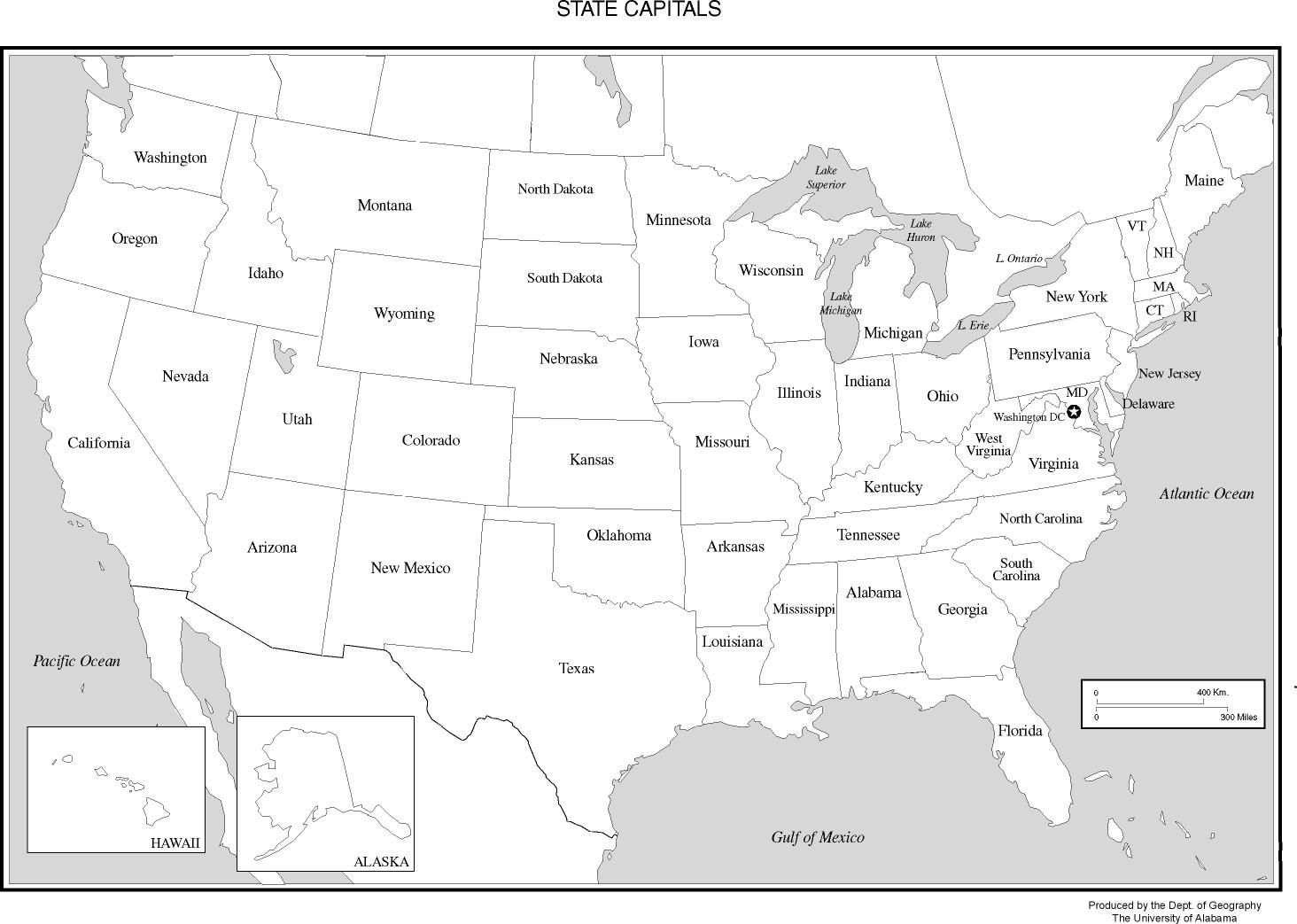
Free Printable Us Map Of States

Use Printable Car Maps To Help Kids Learn Their States On Road Trips

States And Capitals Worksheets State Capitals Worksheet States And

United States Fact Book For Kids
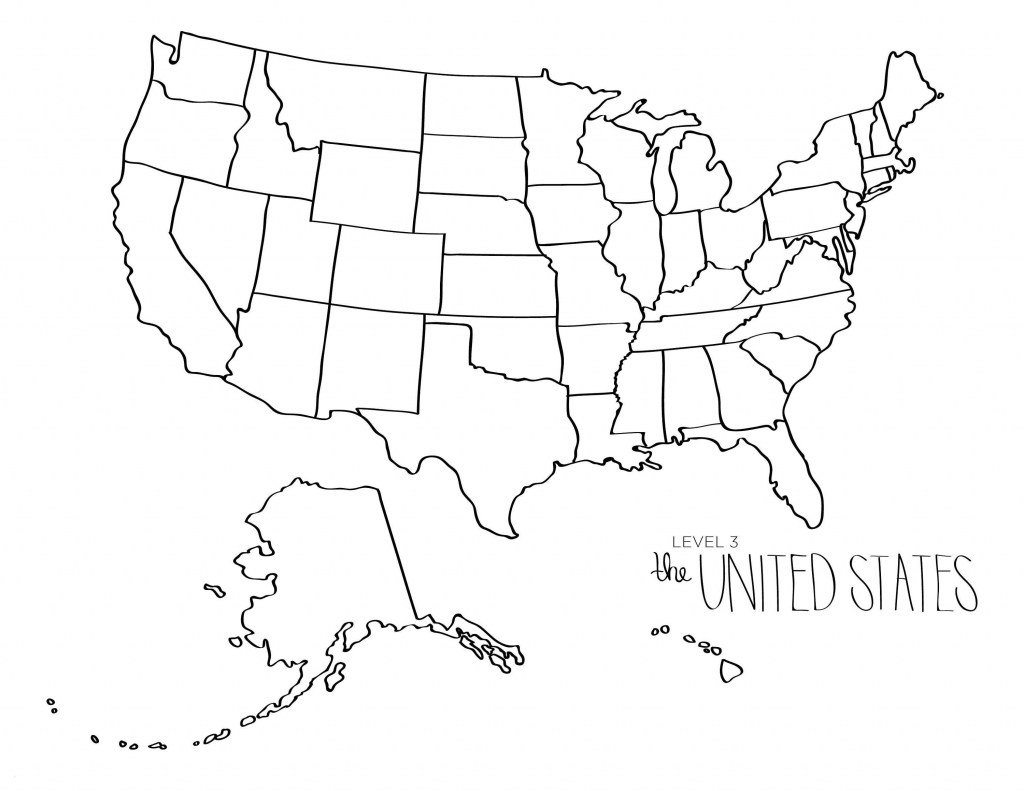
Blank Us Map To Label States 2024 US Map Printable Blank